 
Prescription drugs covered in certain situations Where such a version of a drug does appear on the List of Medications, you will have a 30 day transition period before the exception ceases to apply. For a drug not having a covered generic version.In the case of free doses of a drug pending the approval of a reimbursement by an insurer.For a drug that is not covered by the public plan or a private insurance plan.This form of financial assistance will also be allowed: However, if you change drugs or brands, the exception will cease to apply. If you obtained this type of financial assistance before Apfor a prescription drug, you may continue receiving it for as long as you take this drug. This prohibition applies to any form of financial assistance: copay cards, reductions, reimbursements, free doses, etc. Since April 15, 2021, manufacturers, wholesalers and intermediaries have been prohibited from paying pay you, even partially, for a supply or drug covered under the Public Prescription Drug Insurance Plan or a private plan. In case of a therapeutic failure with at least 2 other drugs biologic drugs used to treat the same medical condition.The request for reimbursement concerns a child or for the remaining duration of their authorization, for a maximum of 12 months following the date of their 18th birthday.A woman is pregnant (including the 12 months following delivery).There is no covered biosimilar version of this drug or until 6 months after the listing of such a version to the List of Medications.Reimbursement of a brand name biologic drug is only authorized in the following cases: 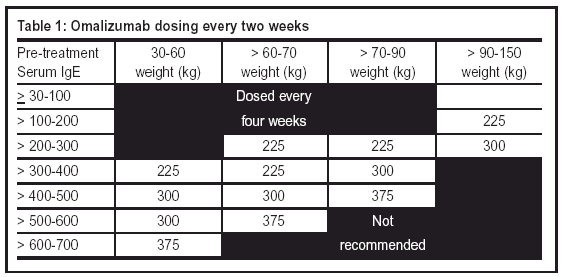
The public plan covers biosimilars specified on the List of Medications. When biosimilars are marketed, the brand name biologic drug to which it is compared is called the reference biologic drug. Biosimilars are very similar copies of biologic drugs, which cost less. Coverage of biosimilar drugsīiologic drugs are drugs produced from living cells and patented by pharmaceutical companies. However, make sure that your new prescriptions still bear the mention “ Ne pas substituer” (after June 1, 2015). Immunosuppressors: Cellcept TM, Myfortic TM, Imuran TM, Rapamune TM, Prograf TM and Advagraf TMįor these drugs, you do not need to obtain a justification from your prescriber.You don’t have to pay the difference between the price of brand name drugs and their generic copies if you obtained them at a pharmacy before June 1, 2015 with the mention “ Ne pas substituer”, even without justification: The drug being prescribed is a brand name drug whose dosage form is essential to obtain the expected clinical results, and this drug is the only one appearing on the List of Medications in this form.You suffer from a documented allergy or intolerance to a non-medicinal ingredient present in the less costly generic name drug, but absent in the brand name drug.The public plan will cover brand name drugs if “ Ne pas substituer” (do not replace) is written on them, along with any of the following justifications: 
Brand name drugs with the mention “ Ne pas substituer” (do not replace) Generics are copies, which cost less they contain the same medicinal ingredients and are approved by Health Canada according to the same quality standards. Difference between a brand name drug and equivalent generic versionīrand name drugs are products that have been patented by pharmaceutical companies. Please note that you will have to pay the difference between the price of a brand name drug and an equivalent generic if you purchase the brand name drug. The public plan covers generics rather than their brand name version if the former is less costly. To find out more, refer to the page Drug Products Undergoing Evaluation and Evaluated on the website of INESSS. The Minister also decides on the replacements for covered drugs that are out of stock. The Minister of Health selects which drugs are covered following consultations with the Institut national d’excellence en santé et en services sociaux ( INESSS). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
